
We are the industry leader in MAM and other MS technologies for HCP, SVA, intact mass, disulfide shuffling, glycosylation, and other CQAs analysis.

Conduct cIEF, CE-SDS, SEC, reverse phase, affinity, IEX & fraction collection followed by in-depth analysis for comprehensive profiling.
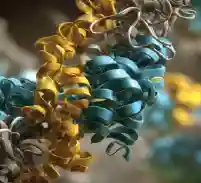
Employ UVCD, DSC, DLS, SEC-MALS, & fluorescence spectroscopy to thoroughly characterize biophysical properties.
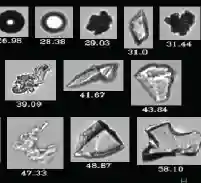
Utilize HIAC, MFI, Flow Imaging Microscopy (FIM) technologies to accurately analyze & quantify particulates, ensuring product purity and safety.
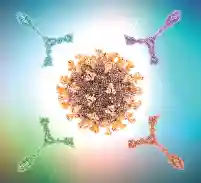
Offer apoptosis-proliferation, cytotoxicity, ADCC, immune checkpoint functional, chemotaxis/cell migration, functional lentiviral vector infectivity/titer assays.

Leverage our extensive experience in frozen, liquid and lyophilization formulation development for formulation screening, stress stability studies, and PQA analysis.

Our modern labs use cutting-edge technology to ensure precise and reliable results. We meet the highest standards, supporting advanced biopharma research and development

As the founder & CEO, I am honored to share our mission of providing top-quality, cost-effective analytical solutions for the biopharma industry. With a background in pioneering the Analytical Platform and MAM at a major biopharma company, I have dedicated my career to advancing analytical excellence.
Our advanced technologies & expert team ensure precision and reliability in every project. We are committed to driving innovation and helping our clients to achieve their goals.
Thank you for trusting BioTherapeutics Solutions as your partner!

Analytical Control Strategy for Biologics. Part I: Foundations
J. Pharm Sci. (2025) 114(7):103826, Kendrick B. S., Sampathkumar K., Gabrielson J. P., Ren D.
Analytical Control Strategy for Biologics. Part II: Roadmap for Development and Implementation
J. Pharm Sci. (2025) 114(8):103834, Sampathkumar K., Kendrick B. S., Gabrielson J.P., Ren D.
Comprehensive multi-attribute method (MAM) workflow for biotherapeutic characterization and cGMP testing
Nat. Protoc. (2023) 8(4):1056-1089, Bones J., Millan-Martin S., Carillo S., Jakes C., Ren D., Rogers R.S.